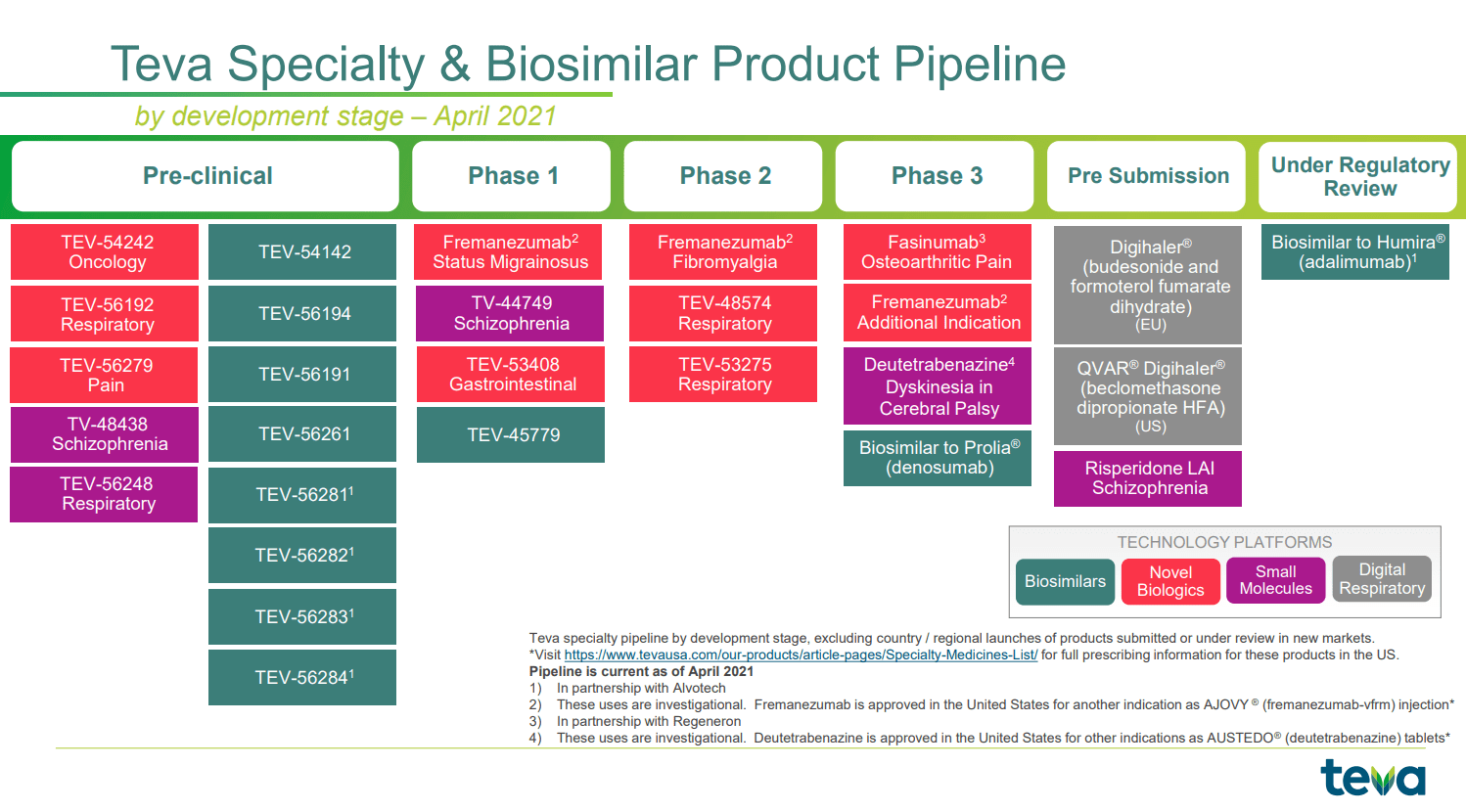
Teva Presents New Long-Term AUSTEDO® (deutetrabenazine) Tablets Data at 2022 American Psychiatric Association ...
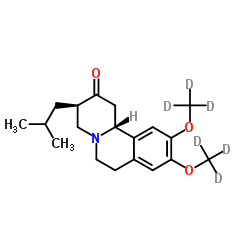
Teva Announces FDA Approval of AUSTEDO™ (deutetrabenazine) Tablets for the Treatment of Chorea Associated with Huntington's Disease - Chemdiv

Teva and Celltrion Launch Truxima (biosimilar- rituximab) to Treat Wegener's Granulomatosis and Microscopic Polyangiitis in the US

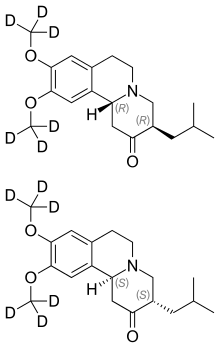
Pharmacokinetic and Metabolic Profile of Deutetrabenazine (TEV‐50717) Compared With Tetrabenazine in Healthy Volunteers - Schneider - 2020 - Clinical and Translational Science - Wiley Online Library

Teva announces FDA approval of Austedo tablets for chorea associated with huntington's disease - Pharma Advancement
Teva Announces FDA Acceptance of Resubmitted New Drug Application for SD-809 for Treatment of Chorea Associated with Huntington Disease - Chemdiv